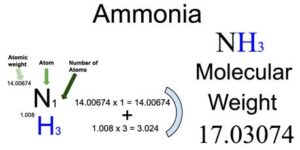
However, your question specifically asks to determine grams of NH3 from grams of H2. Normally, you could decipher between which of the reactants is the limiting reagant by looking at mole ratios, going from molar mass to moles of each species present. You want to remember the mole ratio between each species in the reaction: 1 (N2) : 3 (H2) : 2 (NH3) 
N2 + 3H2 2NH3 which matches the equation you posted. Equations may be balanced by counting the quantity of each type of atom in the reactant and product chemical species, followed by placing numerical coefficients in front of each species until all the atoms are balanced.Īfter balancing your equation, it should appear as follows: One of the first steps in solving stoichiometry questions is to (1) balance the equation. First of all, I was wondering if you could identify what chemistry problem type this question falls under? If you answered a stoichiometry problem, you would be correct! Stoichiometry involves using relationships between reactions & products to figure out some quantitative data in your case, the amount of grams of Ammonia (NH3) produced from the given amount of 6.14g of H2, given the following unbalanced reaction in your question stem: N2 + 3H2 2NH3 Hey there Janeriao! Thank you for posting this question.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
